Clinical development of Rimonabant
Features of Rimonabant
- Name: Rimonabant.
- Therapeutic class: Endocannabinoid type 1 (CB1) receptor antagonist/inverse agonist.
- Route of administration: Oral.
- Current status: In clinical development phase for new therapeutic indications.
- Known security: We have previous efficacy and tolerability data in humans, which facilitates its repositioning.
- Current purpose: To evaluate its effectiveness for the treatment of patients with stroke, spinal cord injury and other pathologies that present fatigue and motor deficits.

Chemical structure of SR141716A (Rimonabant)
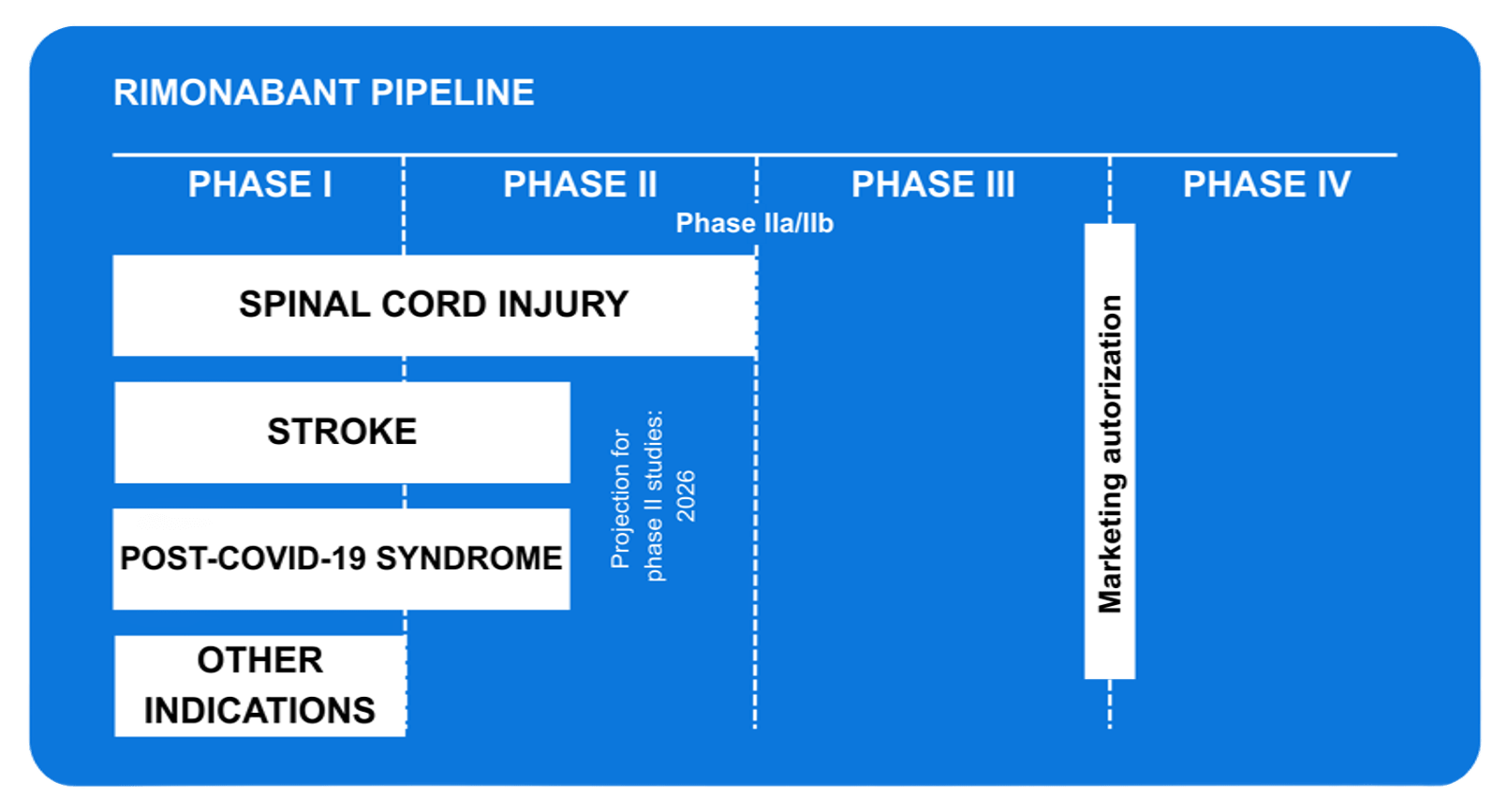
Stage of development
The rimonabant project in Sinfatin is located at:
- Clinical phase
It is currently in the intermediate phase of clinical development, evaluating its efficacy and therapeutic application in phase II and IIb studies. - Priority lines of application:
- Ictus
- Spinal cord injury
- Post-COVID-19 syndrome
- Other possible applications
Potential benefits of Rimonabant
- Modulation of the endocannabinoid system by increasing excitability in motor neurons
- Improvement of motor function after spinal cord injury.
- Known safety profile that facilitates its development for new indications.
Pipeline
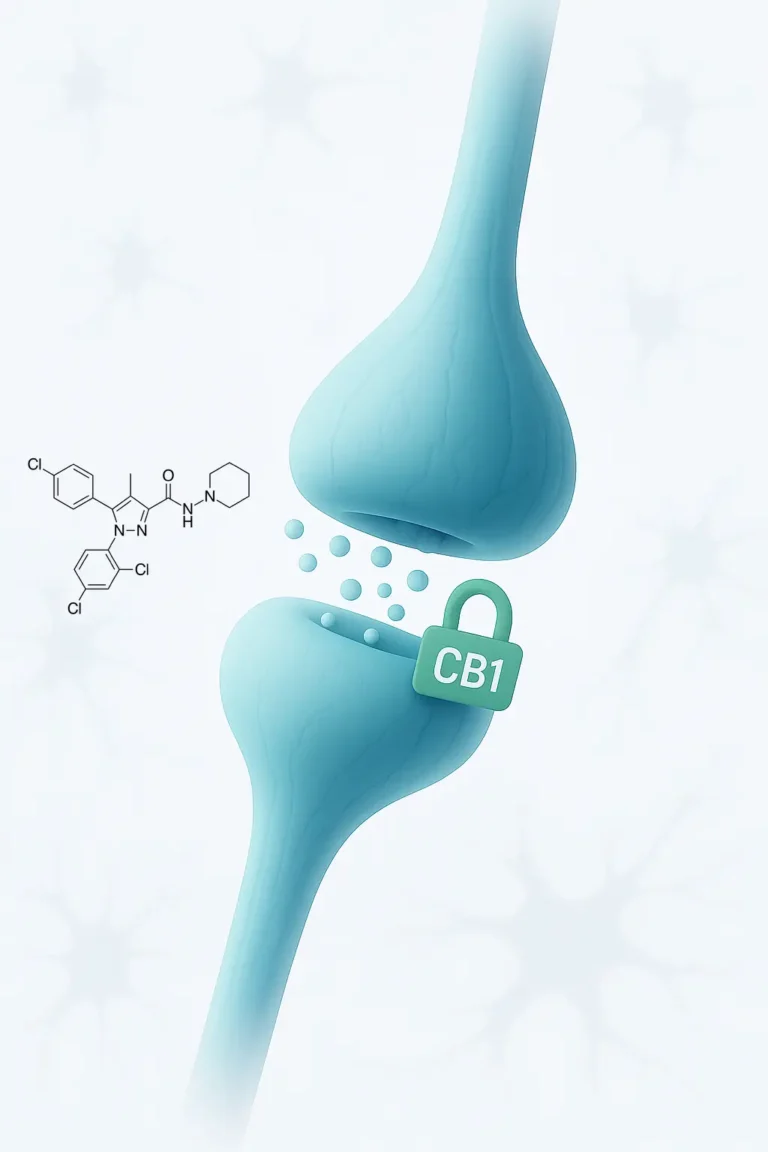
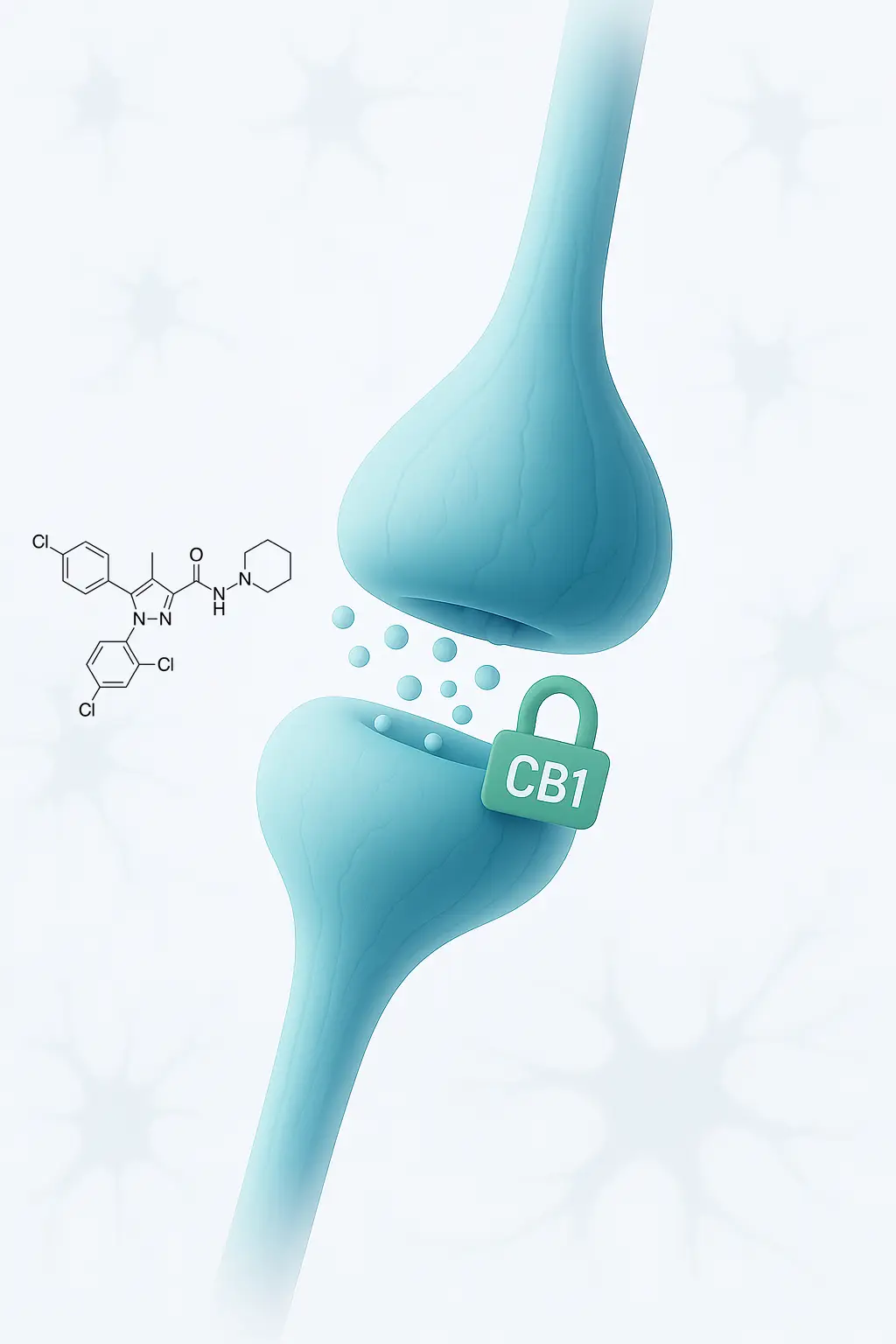
Mechanism of action of Rimonabant
Rimonabant functions as a CB1 receptor antagonist/inverse agonist, primarily targeting receptors located in the central nervous system.
These receptors are an integral part of the endocannabinoid system, which plays a crucial role in regulating neurotransmission.
Normally, endocannabinoids activate CB1 receptors to inhibit neurotransmitter release. However, when rimonabant blocks these receptors, it increases the release of neurotransmitters, especially glutamate.
This action increases neuronal excitability, which is especially beneficial for improving the functionality of the motor system in people who have deterioration resulting from conditions such as spinal cord injuries or strokes.
If you would like more information about this drug, please contact us.
We work to improve the lives of those living with central nervous system disorders by mitigating fatigue due to damage to motor systems.
Our commitment is to move toward more effective, safe, and accessible treatments.
